Monday, 30 March 2015
Is There an Evolutionary Continuity between Spatial Navigation and Declarative Memory?
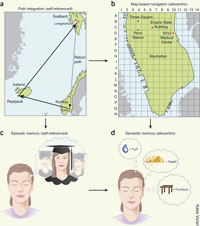
Sometimes someone comes up with a hypothesis whose parts fit together so neatly that it seems amazing that no one has ever thought of it before. A good example is the hypothesis proposed by György Buzsáki and Edvard Moser in the January 2013 issue of the journal Nature Neuroscience, where they propose that there is an evolutionary continuity between the cognitive processes that we use to orient ourselves in space and and the mechanisms that underlie our declarative memory.
Central to this hypothesis is the hippocampus, a brain structure that has long been known to contribute to the storage of memories in human beings. (For example, the famous patient H.M. who had no hippocampus, could not store any new explicit information in long-term memory.) For almost as long, scientists have known that in rats, the activity of the neurons of the hippocampus is closely associated with the animal’s position in space. Buzsaki and Moser’s hypothesis represents one in a series of attempts to reconcile these two seemingly distinct properties of the hippocampus, which displays a certain evolutionary continuity in all mammals.
The first point to note is that to navigate through space, we use two interrelated brain mechanisms that normally work together. The first is called allocentric navigation, which provides static information within an external frame of reference that does not involve ourselves (spatial relationships between markers available in the environment). The second is called egocentric navigation, in which, on the basis of our initial position, the brain constantly calculates distances travelled, turns taken, speed of locomotion, time elapsed, and so on—in short, mentally integrates our path of travel.
The greater or lesser availability of external markers can thus favour one or the other of these two complementary strategies. For example, when it is dark, or in places where there there are few visual markers, the egocentric navigation system has the advantage.
In humans, the hippocampus and the adjacent brain structures involved in spatial navigation are the same ones involved in declarative memory. Declarative memory also takes two distinct forms: semantic memory, which deals with our knowledge of the world at large, and episodic memory, which is our first-person memory of our own life story.
Buzsáki and Moser’s article thus examines how our “travels through space” might be related to the “travels through time” that we make in our declarative memory. As the authors explain, we acquire our semantic knowledge gradually, as we record repeated incidents in our episodic memory, until this knowledge eventually becomes independent of the context. This process is very similar to the way that we develop allocentric maps based on our repeated explorations of our environment (in which the place cells of the hippocampus have been shown to be sensitive to visual markers).
Here is where the parallel proposed by Buzsáki and Moser comes in. They suggest that our semantic memory of the world at large is derived from our allocentric navigation abilities, while our episodic memory of our own biographies is derived from the egocentric navigation skills that we use for our own movements through space. Thus the same networks of neurons may provide algorithms capable of processing both types of travel—spatial and temporal.
It remains to be shown whether a memory system that has evolved from a spatial navigation system could have acquired the vast capacity to store disparate items that is necessary for human semantic memory. This is where the relationship of necessity between the hippocampus and the rest of the cortex when it comes to storing and retrieving memories—a still hotly debated topic—may well come into play.
![]() Memory, navigation and theta rhythm in the hippocampal-entorhinal systems
Memory, navigation and theta rhythm in the hippocampal-entorhinal systems
Memory and the Brain | Comments Closed







